 
An electron is removed from the gaseous sodium to form a sodium cation. The third and fourth steps account for the electron transfer to form ions. Next, diatomic chlorine molecules dissociate into gaseous chlorine atoms. In the first step, solid-state sodium is converted into its gaseous form. The direct route represents the standard enthalpy of formation of NaCl from elemental sodium and chlorine. However, it can be calculated using Hess’s law in a hypothetical series of steps called the Born–Haber cycle, which represents the formation of an ionic compound from its constituent elements.įor example, the Born-Haber cycle for sodium chloride formation considers two alternate routes, one direct and another indirect. In a solid ionic compound, a large number of charged particles interact with each other making it difficult to determine the exact value of the lattice energy experimentally. 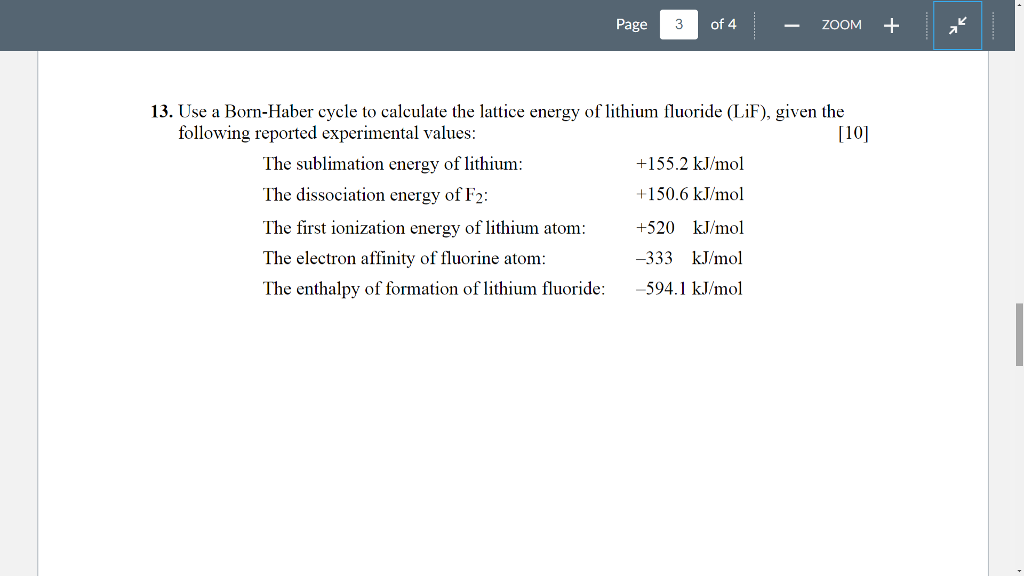
The total energy associated with the formation or breakdown of a crystalline lattice into its gaseous constituents is called lattice energy. The resulting lattice structure is stabilized by decreasing the potential energy, which is released as heat, an exothermic reaction. But where is the energy coming from?Īccording to Coulomb’s law, cations and anions are attracted to each other by strong electrostatic forces into a solid array or lattice. However, when elemental sodium and chlorine react to form a solid sodium chloride crystal, it's a highly exothermic process. Formation of ionic bonds requires an electron transfer from a metal to a nonmetal atom - a process that is often endothermic. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
